|
The combination and optimization of these approaches would have excellent application in point-of-care biodetection to reduce antibiotic misuse, hospitalizations, and deaths from community-acquired pneumonia. Such results obtained with portable devices are promising when compared to non-portable conventional pneumolysin detection techniques such as enzyme-linked immunosorbent assays. Second, magnetic relocation of the particles within the test strip after the immunoassay was completed increased the detected signal by 20%. First, magnetic concentration of the protein before the immunoassay was followed by quantification by means of a mobile telephone camera, and the inductive sensor resulted in detection limits as low as 0.57 ng (telephone camera) and 0.24 ng (inductive sensor) of pneumolysin per milliliter. In this work, a lateral flow immunoassay with magnetic nanoclusters conjugated to anti-pneumolysin antibodies was combined with two strategies to increase the technique’s performance. Spin-orbit interaction r Vl, s µ s Bl r r s Orbiting proton produces a field Bl at the site of the electron. There especially, a rapid diagnostic urine test for pneumococcal pneumonia with high sensitivity and specificity would be helpful in primary care. Pneumonia is the infectious disease that causes the most deaths globally, especially among children under five years and adults over 65 years, most of them in low- and middle-income countries. It is recognized as an important biomarker for diagnosis in urine samples. Pneumolysin is a cholesterol-dependent cytolysin and one of the most important protein virulence factors of pneumonia produced by Streptococcus pneumoniae. This type of detector is easily integrated with the lateral flow immunoassay format. Recently, biofunctional magnetic nanoclusters have been used to selectively label target proteins, which allows their detection and quantification with a magneto-inductive sensor. Improvements in sensitivity would increase their utility in healthcare, food safety, and environmental control. 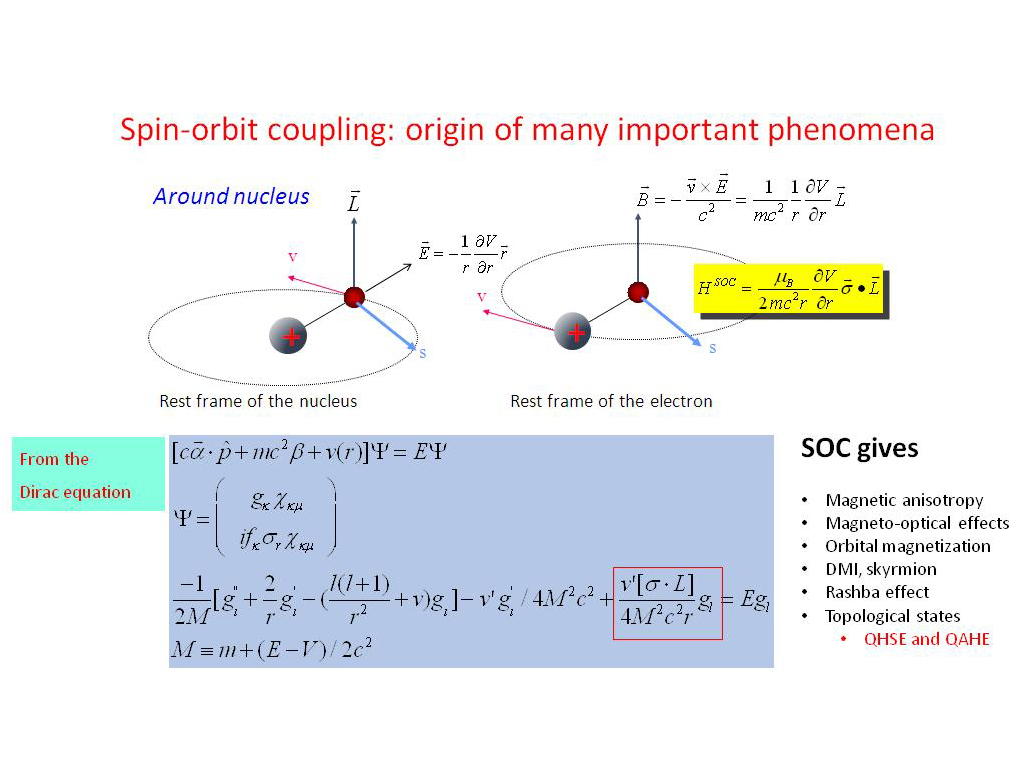
The neutral particles can also show spin orbit coupling since it has both the spin and orbital angular momenta.Lateral flow immunoassays for detecting biomarkers in body fluids are simple, quick, inexpensive point-of-care tests widely used in disease surveillance, such as during the coronavirus disease 2019 (COVID-19) pandemic. The neutrons and protons spin orbital coupling in atom is of great importance as it gives a great contribution towards the total interaction energy The spin orbit coupling of electrons in semiconductor is found have variety of technological applications. The magneto crystalline anisotropy caused due to this interaction. Spin orbital coupling also has many other application. Keywords: spinorbit coupling, Thomas precession, electromagnetic interaction 1. This lead to the splitting in its emission spectra. factorforthespinorbit coupling energy is thus derived without the need to invoke the well-known Thomas precession in the rest-frame of the electron. The electron here has an intrinsic spin given asīoth the spin and orbital momentum couple together and the total angular momentum can be obtained in hydrogen atom. We can observe splitting in hydrogen atom emission spectra due to the spin orbit coupling. (Extrinsic) Spin-Hall Effect Spin Hall Effect: the regular current (J) drives a spin current (J s) across the bar resulting in a spin accumulation at the edges. Here, J represents the total angular quantum number, L gives the orbital quantum number and S represents spin quantum number. We can get the total angular momentum due to the following coupling equation 
The value of this total momentum is maximum when both the spin and orbital momentum is parallel. The total angular momentum is obtained due to the interaction between spin and orbital angular momentum. In simple words we can explain it as how a particles spin and orbital angular momentum interact together. 
This effect is known as spin orbit coupling. This lead to splitting of different energy levels which can lead to different transition energies. 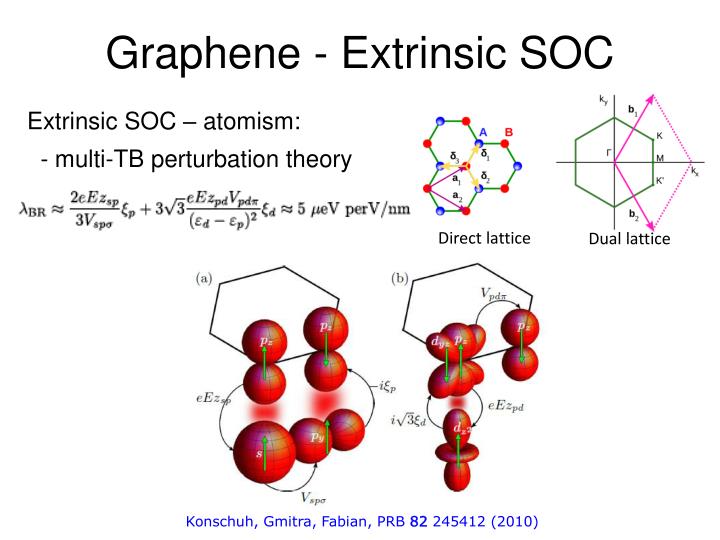
The spin angular momentum of electron can interact with its orbital angular momentum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
